Software for Medical Devices
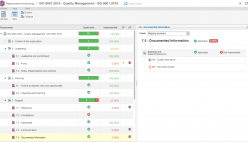
SoftExpert Suite lowers the cost of regulatory compliance and helps companies in the medical devices industry maximize success, increase productivity, reduce risk and adhere to various global regulations such as ISO 9001 and ISO 13485.
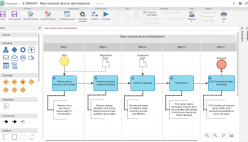
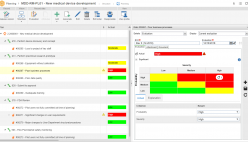
The solution enables companies to continuously improve operations by adhering to various international regulations as well as integrating in a single platform of different management frameworks including: managing risks, maintenance, processes, assets, calibrations, product development, performance indicators, and much more.
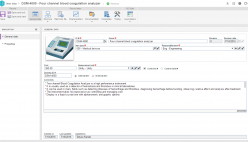
By offering staff with insight in their role in end-to-end activities, they become better able to recognize opportunities improvement and keep up with the dramatic shifts in the industry’s competitive global environment. Powerful support for collaboration with coworkers across organizational boundaries then enables them to play a central role in making smart improvements in the way the organization serves its clients.